
Osmosis is the diffusion of water molecules across a selectively permeable membrane from an area of higher concentration to an area of lower concentration. 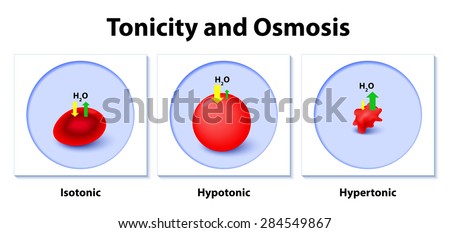
Water molecules will move from the side of higher water concentration to the side of lower concentration until both solutions are isotonic. The hypertonic solution has a lower water concentration than the hypotonic solution, so a concentration gradient of water now exists across the membrane. The hypertonic solution is on one side of the membrane and the hypotonic solution on the other. You now add the two solutions to a beaker that has been divided by a selectively permeable membrane, with pores that are too small for the sugar molecules to pass through, but are big enough for the water molecules to pass through. The second sugar solution is hypertonic to the first. The first sugar solution is hypotonic to the second solution. Solutions of equal solute concentration are isotonic. Last Updated: JanuDefinition Osmolarity is the total solute concentration within a specific volume of a solvent expressed in osmoles per liter (Osm/L) or milliosmoles per liter (mOsm/L). In comparing two solutions of unequal solute concentration, the solution with the higher solute concentration is hypertonic, and the solution with the lower solute concentration is hypotonic. But now you have two mixtures of different solute concentrations. Just like the first cup, the sugar is the solute, and the water is the solvent. Imagine now that you have a second cup with 100ml of water, and you add 45 grams of table sugar to the water. The mixture of a solute in a solvent is called a solution. The sugar dissolves and the mixture that is now in the cup is made up of a solute (the sugar) that is dissolved in the solvent (the water). Imagine you have a cup that has 100ml water, and you add 15g of table sugar to the water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
